CMS recently released the CY 2023 OPPS Proposed Rule, which included proposals for the Hospital Outpatient Quality Reporting Program (OQR) Program. For the full text of the rule, see the Federal Register.
Measures
The OP-31 measure has been a voluntary measure to report but was previously finalized as mandatory beginning with the CY 2025 reporting period. In this rule, CMS proposes to remove the mandatory requirement and continue to make OP-31 a voluntary measure.
Since the removal of OP-26, the Hospital OQR Program does not capture outpatient surgical procedure volume in hospitals. CMS is considering reimplementing OP-26 measure or another volume measure because the shift from the inpatient to outpatient setting has placed greater importance on tracking the volume of outpatient procedures.
Timing of Data Submission
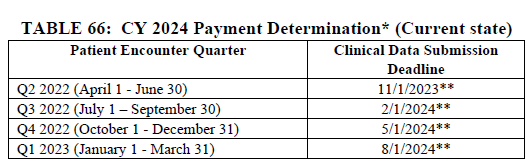
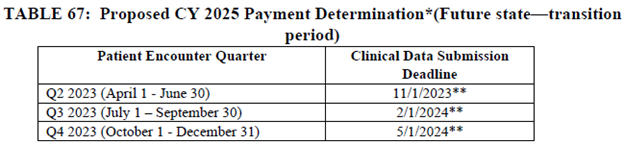
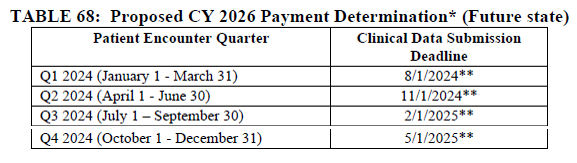
CMS is proposing to align the patient encounter quarters for chart-abstracted measures with the calendar year beginning with the CY 2024 reporting period (CY 2026 payment determination). If finalized, all four quarters of patient encounter data for chart-abstracted measures would be based on the calendar year two years prior to the payment determination year. This proposal will align the patient encounter quarters for chart-abstracted measures with those of the Hospital IQR program.
To transition to this proposed timeframe beginning with the CY 2024 reporting period (CY 2026 payment determination), CMS is proposing to only use three quarters of data for chart-abstracted measures for the CY 2023 reporting period (CY 2025 payment determination).
The following tables were excerpted from the proposed rule:
Validation
For validation purposes, CMS selects a random sample of 450 hospitals and selects an additional 50 hospitals based on targeting criteria. Beginning with validation affecting the CY 2023 reporting period (CY 2025 payment determination) CMS is proposing to add a fifth criterion to the targeting criteria. CMS is proposing that a hospital with less than four quarters of data subject to validation due to receiving an Extraordinary Circumstances Exception (ECE) for one or more quarters and with a two-tailed confidence interval that is less than 75% would be targeted for validation in the subsequent validation year.
If you have questions about reporting your OQR measures, please contact us.

